Principle of Zymography: Zymography is an electrophoresis technique that is used in the detection of protease activity under non-denaturing conditions. It is performed on a zymogram gel, which incorporates the use of a substrate that is copolymerized with polyacrylamide gel. Proteases that catalyze Gelatin , Caesin or Fibrin as a substrate will show up as clearings against a dark blue background after staining with Coomassie brilliant blue.
The use of zymography encompasses the use of many different buffer systems. Sample buffer is used in conjunction with the protein sample for a few purposes. Sample buffer helps to control pH, controls sample/ion movement, increase sample viscosity and density (aid in loading into sample wells), provide tracking dye (monitor seperation during electrophoresis), provide denaturing molecules (SDS to linearise proteins) and provide chemical reducing agents (control sample chemistries). Another buffer system is the running buffer system. Sample wells are filled with running buffer which contains ions that helps in the constant migration of proteins towards the bottom of the well, when voltage/current is applied. The 3rd buffer system that is used is the renaturation buffer system which helps to renature (allows protein to fold back) to their tertiary structure in order for it to exhibit enzymatic activity on the gelatin substrate of the zymogram gel. Proteins in its linearised state do not exhibit enzyamtic activity (linearised by SDS) and requires renautration using renaturation buffer to restore its enzymatic activity. The fourth buffer system that is used is the developing buffer system which is used to develop the zymogram gel after addition of renaturation buffer. Developing buffer allows protease to exhibit enzymatic activity on the zymogram gel (digest gelatin).
The gel is stained with Coomassie Brilliant Blue stain (Biosafe Biorad) to visualise the protease activity, which will stain the whole zymogram gel blue in colour. The zymogram gel is then destained with MilliQ water to remove excess commassie blue and proteins that exhbit enzymatic activity will produce clearings against a dark blue background. Coomassie Brilliant Blue stain has high affinity to proteins present on the 10% gelatin zymogram gel. Gelatin is considered a protein substrate as well as the periplasmic proteins that is present on the zymogram gel after seperation by electrophoresis. Coomassie Brilliant Blue stain binds tightly to both of these proteins, hence, the gel appears blue in colour. Periplasmic proteins which exhibits protease enzymatic activity on the gel, will digest the gelatin substrates. Hence the region of the zymogram which is digested lacks proteins. Without proteins, Coomassie Brilliant Blue stain will have poor affinity with the gel and can easily be destained/washed off in the presence of destaining solution/DI water.
1D-Zymography is an electrophoresis technique that seperates proteins 1 dimensionally based on molecular weight on the zymogram gel. Proteins are linearised with SDS and carry an overall negative net charge. Larger proteins have more SDS bound to it and smaller proteins have less SDS bound to it. This ensure that proteins have a constant mass to charge ratio of 1.4g SDS/protein. When an electric current is applied, proteins will migrate towards the anode (+) of the gel. Larger molecular weight proteins will migrate slower and appear at the bottom of the gel. Smaller molecular weight proteins migrate faster and appear in higher regions of gel. Proteins that exhibit enzymatic activity on the gelatin substrate, will appear as clearings against a dark blue blackground after destaining in the destaining solution.
Methods
Sample Preparation
1. Add/pipette in 10 ul of SDS-Sample buffer into each of the 11 eppendorf tubes ( 11 tues for 11 isolated)
Reason: Equal volume of sample buffer added to equal volume of sample is important, as it allows equal amount of SDS to bind to equal amount of proteins to have a constant mass to charge ratio of 1.4g SDS/ protein.
2. Add/Pipette in 10ul of Sample + DI water (periplasmic proteins) into each of the 11 eppendorf tubes
Reason: DI water is added to dilute the samples if concentration is deemed to be too high
The amount/volume of periplasmic proteins are to load, are calculated by dividing the mass of protein (constant fixed at 10ug) with the concentration of protein obtained using Bradford assay. DI water is used to top up the remaining volume to 10ul. Total volume of sample + sample buffer + DI water should be 20 ul. 20ul is the maximum capacity of well
3 Centrifuge the eppendorf tubes for 7 seconds at 16 000xg
Reason: This is to prevent air bubbles from forming during pipetting and ensures homogenous mixing of sample with the sample buffer
Prepare Running Buffer
1. Add 100ml of TGS running buffer (10X) to 900ml of DI water using a measuring cylinder
Reason: This is to dilute the TGS buffer 10X. It is more practical to have 1 bottle of concentrated TGS rather than 10 bottles of diluted TGS buffer as it saves space
Assembly of Xcell SureLock Mini-Cell
1. Fill Upper and Lower Buffer Chamber with 1X TGS running buffer
Reason: TGS running buffer contains ions for migration of protein when applied to an electric field
2. Gently remove the gel comb and peel tape off from gel cassette
3. Insert the gel comb into the Xcell SureLock Mini-Cell and lock it into place using the Gel Tension Wedge.
4. Load 20ul of sample into the each of the wells (* Load from 2nd well onwards as 1st well is for the protein ladder )
5. Load 8.0ul of protein ladder into the 1st well of the Zymogram gel
Reason: Protein ladder helps to track the migration of protein during electrophoresis
6. Close cover of the Xcell SureLock Mini-Cell
Reason: Ensures that electrophoresis can begin
Electrophoresis
1. Switch on the power supply and ensure that the electrodes are connected
2. Run the gel at 120 V for 90 minutes ( The optimal voltage for running the gel is 120 volts. Running the gel at very high voltages (e.g. 200 volts) leads to the generation of intense heat, which may denature the proteins. Running at too low voltages may result in long duration before electrophoresis is complete. )
Removal of gel after electrophoresis
1. Unlock the Gel Tension Wedge and remove the comb from the Xcell SureLock Mini-Cell
2. Use the gel knife to separate the plates and extract the 10% Zymogram gel and placed on plastic tray
3. Rinse the gel with DI water
Reason: To wash away the TGS running buffer and SDS
4. Add 100ml of renaturation buffer (contains Triton-X and detergent) and incubate with gentle agitation for 30 minutes
Reason: This is to allow proteins to renature back to its teritiary structure
5. Decant the renaturation buffer
6. Add 50ml of Developing buffer and incubate with gentle agitation for 30 minutes
Reason: This is to allow proteases to exhibit enymatic activity on the Zymogram by digesting the gelatin
7. Decant the Developing buffer
8. Add 50 ml of Developing buffer and incubate at 37°C overnight for 20 hours
Reason: This is to allow proteases to exhibit enymatic activity on the Zymogram by digesting the gelatin. The purpose of developing twice is to allow the proteins to adapt to the gradual change from renaturation to developing buffer.
9. Decant the Developing buffer
10. Add Biorad Biosafe Coomassie Blue G-250 stain and incubate with gentle agitation overnight
Reason: Ensure that the Zymogram gel is fully submerged in the stain. Biorad Biosafe Coomassie Blue G-250 stains the entire Zymogram gel blue in colour
11. Decant Biorad Biosafe Coomassie Blue G-250 stain
12. Add MilliQ water (destaining solution) and incubate with gentle agitation overnight
Reason: This is to destain the gel and only areas that protease have exhibited enzymatic activity on the gel will appear as clear bands against a dark blue blackground
Timescale
The whole proccess of running a 1D-zymogram takes about 3 days. 1 day is attributed to the actual electrophoresis, 1 day for staining and another day for the destaining proccess.
The results of a 1D-zymogram
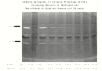
Picture is taken from: astro.temple.edu
The quantity and size of clearings (transparent bands) indicate the amount of protease enzymatic activity on the gelatin substrate of the gelatin gel. S. maltophilia of different isolates can be screened using this methodology to determine which isolate produce the most interesting (most quantity and largest clearings) on the 1D-Zymogram. This isolate can then be further analysed using a 2D-Zymogram.
Sorry for the long entry guys, hope it won't bore you. Thanks alot, and enjoy the last 2 weeks!
From: Benjamin Ma Xianwei
Class: TG01
0606181F
12 comments:
Hello.
Got some questions.
What is the zymogram gel made up of? And why must you denature and later renature the proteins?
Thanks =).
Li Ping
0607498C
TG o2
Hi Benjamin,
Is it a must to use MiliQ water or can DI water be used?
And how is Zymogram gel different from the normal polyacrylamide gel?
For low voltage used during electrophoresis, other than the long duration, does it affect the results?
Thanks
Ting Ying Chee
TG01
Hi Li Ping
A zymogram gel is made up of resolving gel and stacking gel. It contains acrylamide, bis-acrylamide, ammonium persulfate, TEMED, gelatin substrate, SDS and water. For normal SDS-PAGE it is important to denature the protein from its tertiary structure into its primary structure. THis is to allow proteins to be linearised so that SDS can bind and produce a constant mass to charge ratio for all the proteins regardless of its size (all proteins become negative charge) => hence allowing the seperation of proteins by its molecular weight. For Zymogram, its abit special as after SDS-PAGE is completed, there is an extra renaturation step. This renaturation step is to fold proteins and proteases back from its linearised state back to its original conformation. Proteases must be in its original conformation (active site specific to substrate) in order to digest gelatin. If the proteases are still in linearised state, its active site is not specific to gelatin substrate and unable to digest it and produce clearings on the 1D-Zymogram => thus, the reason for renaturation step
From: Benjamin Ma
TG01
0606181F
Hi Ying Chee
For destaining, we use MilliQ water to destain as it removesthe unbound coomassie blue stain quite easily. MilliQ water has higher purity than DI water so MilliQ water is prefered.
A zymogram gel is used for the detection of protease activity and contains a substrate for the proteases to digest. A normal polyacrlamide gel is for seperation of proteins and not to detect protease activity, hence it does not contain a substrate. Otherwise, zymogram and polyacrylamide gel are the same.
A low voltage will just result in slower seperation of proteins. As such proteins in the upper region of the gel may be susceptible to diffusion while seperation is still taking place. It has been found that 120V is optimal for 1D-Zymogram seperation.
From: Benjamin Ma
Class:tg01
0606181F
Hi Ben,
In ur entry, u mentioned that the zymogram gel is then destained with MilliQ water to remove excess commassie blue and proteins that exhbit enzymatic activity will produce clearings against a dark blue background. So the milliQ water just remove the excess stain?If so, why does the excess stain get remove while the 'non-excess' doesn't?
Sorry, but wad id TGS running buffer?
Dyana
0605169B
Hi Dyana,
Sorry for the misconception. I will start with the staining procedure whereby the Zymogram gel after 1D-seperation, is stained with Coomassie Brilliant Blue stain. During this staining step, it is important for the whole gel to be completely submerged in the stain. This is because Coomassie Brilliant Blue stain have high affinity to proteins (gelatin and the periplasmic proteins are proteins) and stain the whole gel blue. These proteins on the gel get bound to the protein. Those Coomassie Brilliant Blue stain that is unbound to the protein are termed as excess (because the stain is added in volume that can submerged the gel). Thus after 1day, during the destaining proccess, MilliQ water will wash away this unbound Coomassie Brilliant Blue stain (not bound to gelatin or periplasmic proteins, non-excess) => and this unbound region can only be regions whereby protease activity have digested the gelatin => thus gelatin cannot bind to Coomassie Brilliant Blue stain and produce clearings.
TGS running buffer is Tris-Glycine running Buffer
Thanks!
From: benjamin Ma xianwei
tg01
0606181F
Hi benjamin,
What happens if the excess commassie blue isn't removed? Will it affect the staining and the outcome of the gel when you visualise it?
Is it commassie brilliant blue the only reagent that can be used for staining the zymogram gel?
Thanks.
Lyn
TG02
hi ben,
u mentioned that the isolate can then be further analysed using a 2D-Zymogram. Since 1D-Zymogram is able to show the amt of protease enzymatic activity, i'm curious what will the 2D-Zymogram show? thanks.
Malerie
TG02
Hi Lyn,
The remaining excess coommassie blue will eventually be removed during the destaining step which is to decant the stain and add destaining solution: MilliQ water after 1 day of staining. If the staining takes longer than 1day, the whole gel will blue in colour, same as after staining in 1 day, and does not make any difference. This is because if the gel contains proteins (gelatin and periplasmic proteins) it will bind to the stain and not be seperated. THus, even if you stain longer or add the stain in excess, all the stain will have bound to all the proteins already, it will not make the ZYmogram any bluer in colour. Thus, it is important to cover the whole gel in the commassie blue stain during the staining step to ensure all proteins are bound to the stain ad remove them during the destaining solution.Coomassie blue is the only stain for zymogram gel as it has high affinity to proteins
Thanks!
From: Ma xianwei Benjmain
class;tg01
0606181F
Hi Malarie,
Please refer to Han Yang's post entry for more information on 2D-Zymogram
From: Benjamin Ma Xianwei
Class: tg01
0606181F
Hello, I was wondering if I do a normal SDS-PAGE gel and then a zymography, with the same sample, if any proteins with activity were present in my sample, I would be able to see them stained with coomassie on the regular gel anyway, just would not be able to tell if the possess activity or not, am I correct in my assumption? they would stain on the regular gel, and I would then see their activity on the zymography, right? thank you!
Hello,
does someone know how(long) I can store my sample in sample buffer ?? Thank you !!
Nicole
Post a Comment